
Buying guides
Pulsetto vs Truvaga: which vagus nerve stimulator actually delivers in 2026 (honest comparison)
Pulsetto and Truvaga both cost $250–$270 and target the same vagus nerve. Here's the honest head-to-head — app vs. clinical lineage, who should buy which.
“Transcutaneous vagus nerve stimulation has promising results across small studies, but the field needs larger, sham-controlled trials before any device should be sold as a depression or anxiety cure.”
You've already decided non-invasive vagus nerve stimulation is worth a try. You've narrowed it to two devices: Pulsetto at ~$270, with the slick app and the TikTok ad you keep seeing, and Truvaga at ~$250, with the quieter marketing and the FDA-510(k) family tree. They share roughly the same price band, the same target nerve, and 95% of the search results overlap — but they are not the same purchase.
Most "Pulsetto vs Truvaga" pages online are either affiliate roundups that conveniently bury one device or Reddit threads with one anecdote apiece. This is the version that actually puts the two side by side, names the trade-offs, and tells you when neither is the right buy.
The two-minute verdict
| Pulsetto | Truvaga | |
|---|---|---|
| Price (hardware) | ~$270 | ~$250 |
| Form factor | Neck-worn collar, hands-free | Handheld, press to neck |
| Session length | 4–20 min, mode-dependent | ~2 min per side, repeatable |
| FDA status | Wellness device | 510(k) lineage (gammaCore family) |
| App | Broad protocol library (anxiety, sleep, focus, burnout, pain) | Minimal companion app |
| Subscription | Premium tier gates advanced protocols | None |
| Mechanism | Transcutaneous cervical-branch tVNS | Transcutaneous cervical-branch tVNS |
| Best for | Daily-ritual users who want guided protocols | Users who trust clinical pedigree over UX |
| Woo-Woo Meter | 3/5 | 3/5 |
If you only have thirty seconds: pick Pulsetto if you'll use the app daily; pick Truvaga if a 510(k)-cleared lineage matters more to you than features. If you don't know which sentence describes you, the rest of this post is for you.
What both devices actually do (and don't)
"The tVNS field is promising but no consumer device should be marketed as a depression or anxiety cure until the trials are larger and sham-controlled."
Both are transcutaneous vagus nerve stimulators. They send a small electrical pulse through the skin of the neck to stimulate the cervical branch of the vagus nerve — the same anatomical target the clinical literature has been probing for migraine, treatment-resistant depression, and anxiety. The mechanism is plausible and shared. Where they differ is form factor, software, and regulatory standing.
The evidence picture is the same for both, because the mechanism is the same. The Yap et al. 2020 review in Frontiers in Neuroscience is still the cleanest summary: dozens of small RCTs across anxiety, depression, and stress endpoints; signal is there; protocols are inconsistent; trials are too small and too short to be conclusive. The autonomic-balance rationale — that improving vagal tone improves HRV and parasympathetic state — has its own foundational HRV literature, and that part is solid. The leap from "shared mechanism" to "this specific consumer device will treat your anxiety" is the leap neither company should be making, and both companies' marketing makes anyway.
Neither device is FDA-cleared as an anxiety or depression treatment. Truvaga is sold by Electrocore, the company behind gammaCore — a prescription tVNS device with 510(k) clearances for cluster headache and migraine. Truvaga shares the clinical lineage but is sold as a consumer wellness device. Pulsetto has no comparable regulatory pedigree; it's a direct-to-consumer wellness device from the start.
For the longer evidence walkthrough, see our vagus nerve devices evidence breakdown and the wider best vagus nerve devices 2026 roundup.
Pulsetto: strengths, weaknesses, who it's for
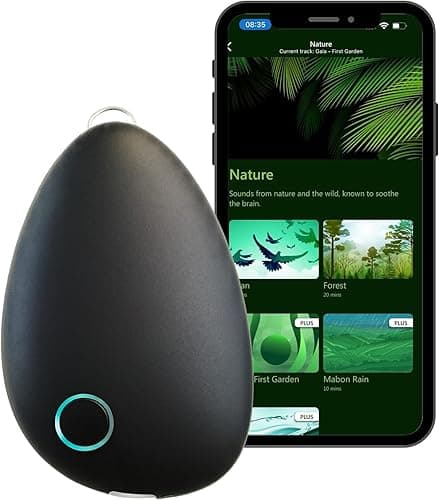
Pulsetto is the experience-led device. The collar sits around your neck, two electrodes contact the cervical branches on either side, and you start a session from the app: anxiety, sleep, focus, burnout, pain — each is a different stimulation pattern, typically 4 minutes. It's hands-free. You can lie down, sit at your desk, or do it on the couch.
What it does well. The app is the real product. The protocol library is broad, the sessions are short enough to actually do daily, and the hands-free form factor means people stick with it. Adherence is the single biggest predictor of any benefit from this category, and Pulsetto wins on adherence by a wide margin.
What it doesn't do well. The premium subscription gates advanced protocols, which feels gratuitous on a $270 device. Marketing is louder than the evidence justifies — "rewire your nervous system" is in the copy somewhere, and that overshoots what tVNS has been shown to do. Electrode placement on a collar is approximate; some users never quite find the spot that produces a clean sensation.
Pulsetto is for the buyer who wants a daily ritual, who likes guided protocols, and who knows about themselves that an app keeps them honest. Our full Pulsetto honest review covers the longer take, and our Pulsetto vs Sensate comparison explains why we don't think the two are the same category at all.
Truvaga: strengths, weaknesses, who it's for
Truvaga is the clinical-feeling device. A handheld unit with two contact points; you press it to one side of your neck for two minutes, then the other side. There's a minimal companion app, but the device works without it. No subscription. The button-press UX is closer to a prescription medical device than a wellness gadget — which is, of course, the family it comes from.
What it does well. Pedigree. Truvaga is in the gammaCore product line, and gammaCore is the device that built the clinical tVNS literature. The unit feels engineered; the stimulation is consistent; the protocol is simple enough to follow without thinking. No subscription means the price you pay is the price you pay.
What it doesn't do well. The experience is sparse. There's no broad protocol library, no anxiety-mode-vs-sleep-mode framing, no progress visualizations. For users who need software hand-holding to keep a habit, Truvaga is a worse fit. Holding a device to your neck for four minutes (two per side) is harder to do during a meeting than wearing a collar; harder to do hands-free in bed.
Truvaga is for the buyer who trusts a 510(k) lineage more than an app, who doesn't want a subscription, and who'd rather the device get out of the way once they've learned the protocol.
Direct head-to-head: the dimensions that actually matter
Comfort. Pulsetto wins for sessions over 5 minutes — a collar is more comfortable than holding a device to your neck. Truvaga wins for sessions under 3 minutes — there's nothing to wear, nothing to adjust.
Daily friction. Pulsetto wins. Hands-free + app reminder beats handheld + manual two-side routine on adherence. This is the most important dimension.
Evidence strength. Tie on mechanism. Truvaga wins on regulatory lineage; the consumer product is not the cleared one, but the family tree is closer to the published clinical work.
App and protocol depth. Pulsetto wins, decisively. Pulsetto loses, equally decisively, on subscription gating.
Total 12-month cost of ownership. Truvaga is cheaper if you don't subscribe to anything ($250 flat). Pulsetto is roughly equivalent if you stick to the free tier, more if you take the premium subscription. Check current Pulsetto subscription pricing on the product page; it shifts.
The conversation with your therapist or doctor. Truvaga is easier to mention — the 510(k) lineage gives clinicians something to anchor on. Pulsetto reads as a consumer wellness device, full stop.
Refund / return reality. Both have direct-from-brand trial windows and Amazon's standard return policy. Both need 4–8 weeks of daily use to fairly judge, which means the trial window matters more than a few dollars of price. Read the current terms at checkout — they change.
Use-case picks
- For anxiety, daily maintenance: Pulsetto. The app's anxiety protocol slots into a morning routine; adherence carries the benefit. The evidence base is the same as Truvaga's, but the device you actually use is the one that helps.
- For sleep onset: Pulsetto. Hands-free during a wind-down is the right form factor for bedtime. The sleep protocol is designed for it.
- For users with a migraine history who want a clinically-adjacent option: Truvaga, but talk to a neurologist first. gammaCore is the cleared option for migraine; Truvaga is the consumer cousin, not the cleared device. Do not self-treat migraine with a wellness-labeled product without medical input.
- For users who hate subscriptions on principle: Truvaga. $250 once, done.
- For users who'd benefit more from biofeedback than stimulation: Neither. HeartMath Inner Balance shows you HRV-driven coherence in real time and teaches a transferable breathing skill — see our biofeedback for anxiety roundup and the HeartMath honest review.
- For users whose anxiety lives in rumination, not autonomic state: Neither. Muse S Gen 2 addresses attention training, which is a different mechanism than vagal stimulation.
- For users who want passive all-day nervous-system nudging: Neither. Apollo Neuro is a wearable, not a session device — different category, different trade-offs, covered in our Apollo Neuro honest review.
What neither device will do
Neither will replace therapy. Neither will resolve a sleep disorder you've ignored for two years. Neither has the trial base to be sold as a treatment for major depression. Neither belongs at the front of your stack if you haven't tried the free interventions first: paced breathing at six breaths per minute, daily walks, sleep regularity, an actual therapist. tVNS is an adjunct to those, not a substitute.
If you have a history of cardiac arrhythmia, an implanted electrical device, vagal nerve damage, or severe sleep apnea, talk to a clinician before buying either. The published caveats in the tVNS literature are narrow but real, and a $250 wellness device with no medical supervision is not the place to learn about them.
Verdict and Woo-Woo Meter
Both devices land at 3/5 on the Woo-Woo Meter: real mechanism, real but emerging evidence, marketing that outruns the data on both sides. For the rubric behind the rating, see what is the Woo-Woo Meter.
Pick Pulsetto if you want the app-driven daily ritual, you'll actually use the protocol library, and the hands-free form factor matches your life. Budget for the subscription if you want the broader protocols.
Pick Truvaga if you weight regulatory pedigree higher than app features, you want a no-subscription device, and you prefer a handheld you can stash in a drawer when you're not using it.
The honest meta-point. The difference between these two devices is smaller than the difference between using one consistently for 8 weeks and buying it, doing two sessions, and putting it on a shelf. Adherence is the single biggest variable in this category. Pick the one you'll actually use daily — and if you can't honestly answer that question, you don't yet need either device.
Products mentioned in this post

Pulsetto Vagus Nerve Stimulator
App-controlled non-invasive vagus nerve stimulator for stress, sleep, and anxiety.

Truvaga Vagus Nerve Stimulator
Handheld clinical-grade vagus nerve stimulator — quick daily sessions.

Apollo Neuro Wearable
Vibration-based wearable for stress relief and sleep, with growing clinical evidence.
Sensate 2 Relaxation Device
Vagus nerve relaxation device using infrasound vibration for stress and anxiety relief.

HeartMath Inner Balance Sensor
HeartMath's HRV biofeedback sensor for coherence and resilience training.

Muse S Gen 2 Headband
Soft fabric EEG headband for both meditation and overnight sleep tracking.
Frequently asked
- Is Truvaga the same as gammaCore?
- No. gammaCore is the prescription tVNS device from Electrocore with FDA 510(k) clearances for cluster headache and migraine. Truvaga is a separate consumer wellness device from the same company; it shares the mechanism and engineering lineage, but it's not the FDA-cleared product. Don't conflate them. If you need the cleared device for a diagnosed condition, ask a neurologist about gammaCore — not Truvaga.
- Is Pulsetto FDA-cleared?
- No. Pulsetto is sold as a wellness device, not an FDA-cleared medical device. Some consumer vagus nerve stimulators in this device class have 510(k) clearances — Pulsetto isn't currently among them. This doesn't make the mechanism unreal; it does mean Pulsetto cannot be marketed or used as a treatment for anxiety, depression, or any other diagnosed condition.
- Will I feel anything during a session?
- Yes — both produce a noticeable tingling or buzzing sensation along the side of the neck, and most users titrate the intensity up over the first week as the novelty fades. Mild jaw tension, chin tingling, or hoarseness during a session are common and resolve when you stop. If you feel sharp pain or dizziness, you're either at the wrong intensity or the electrode placement is off; reposition rather than push through.
- How long before I know if either device is doing anything?
- Acute calming effects are immediate for some users — a noticeable shift in breathing or muscle tension by the end of the first session. Durable changes in baseline anxiety, HRV, or sleep take 4–8 weeks of near-daily use, and even then they're confounded by sleep, exercise, alcohol, and life. Anyone promising results in a week is selling, not informing.
- Does insurance or HSA/FSA cover Pulsetto or Truvaga?
- Insurance: effectively never. Both are sold as wellness devices, not prescribed medical devices, and that's not a reimbursable category. HSA/FSA coverage is plan-specific — some administrators accept either device with a letter of medical necessity, most don't. Check with your administrator before assuming either qualifies.
Sources
- [1]Critical Review of Transcutaneous Vagus Nerve Stimulation: Challenges for Translation to Clinical Practice · Frontiers in Neuroscience · 2020-04-28
- [2]510(k) Premarket Notification — Non-invasive Vagus Nerve Stimulator · U.S. Food and Drug Administration · 2017-04-18unverified
- [3]An Overview of Heart Rate Variability Metrics and Norms · Frontiers in Public Health · 2017-09-28
- [4]Amazon product listings (current pricing) · Amazon.com · 2026-04-09
Related reading
More from the desk

Buying guides
Best Vagus Nerve Stimulation Devices in 2026: Honest Rankings
We ranked the top vagus nerve stimulation devices for 2026 by evidence, build quality, and value. Honest picks with Woo-Woo Meter ratings for each.

Buying guides
Biofeedback for anxiety: which devices actually work in 2026 (an honest review)
Honest 2026 guide to biofeedback devices for anxiety — HeartMath, Muse 2, Pulsetto, Sensate, Apollo Neuro. Real evidence, real prices, Woo-Woo Meter ratings.

Buying guides
Pulsetto vs Sensate: which "calm your vagus nerve" device actually delivers?
Pulsetto and Sensate both promise a calmer nervous system but work on different mechanisms. Here's the honest side-by-side — and who should buy which.